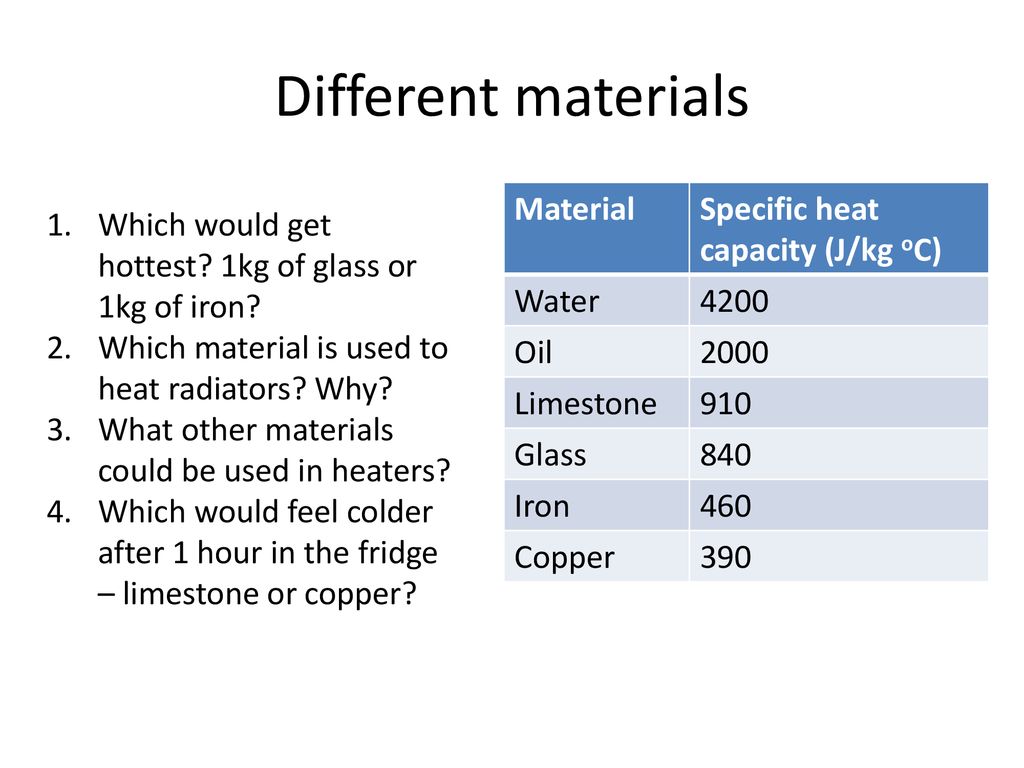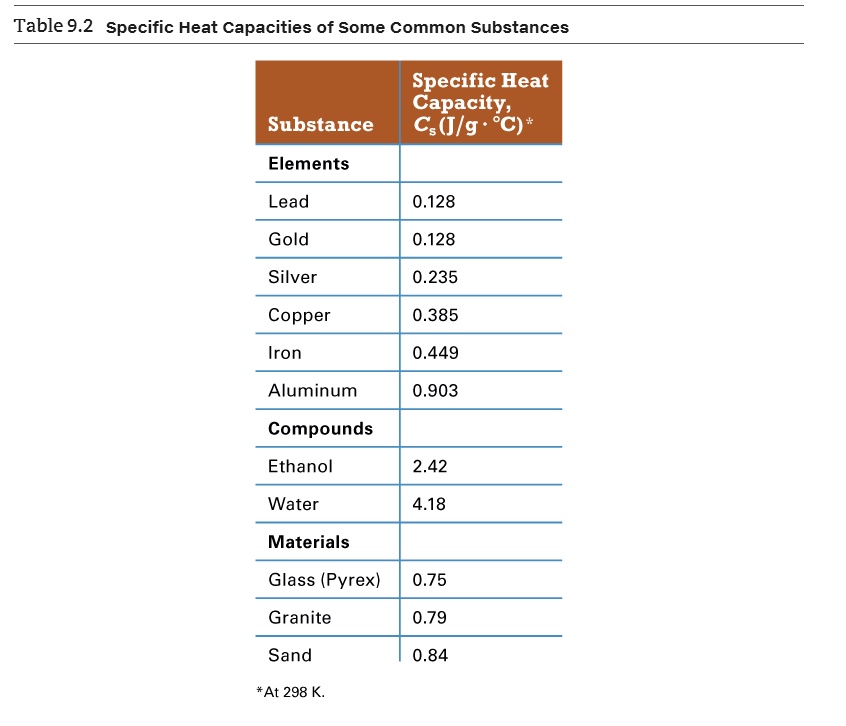
SOLVED: Table 9.2 Specific Heat Capacities of Some Common Substances Specific Heat Capacity; Cs(Jlg. *C) Substance Elements Lead 0.128 Gold 0.128 Silver 0.235 Copper 0.385 Iron 0.449 Aluminum 0.903 Compounds Ethanol 2.42

SOLVED: Aheat energy of 645 J is applied to a sample of glass with a mass of 28.4g: point Its temperature increases from -11.6 'C to 15.5 'C. Calculate the specific heat

Volumetric heat capacity enhancement in ultrathin fluorocarbon polymers for capacitive thermal management | Semantic Scholar

Unveiling the Dependence of Glass Transitions on Mixing Thermodynamics in Miscible Systems | Scientific Reports

.jpg)
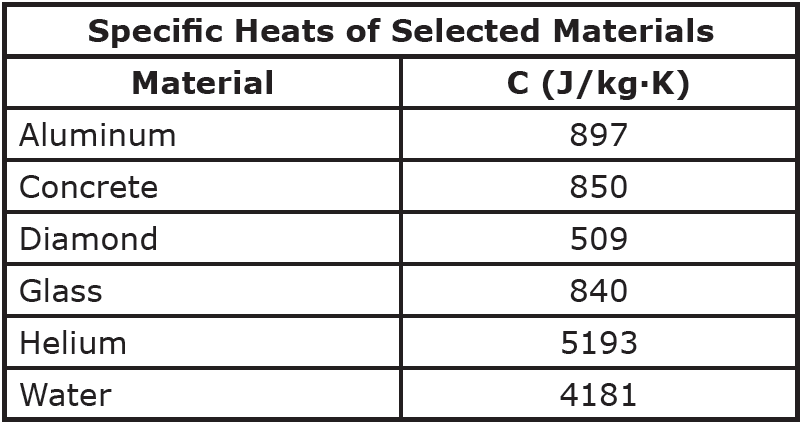
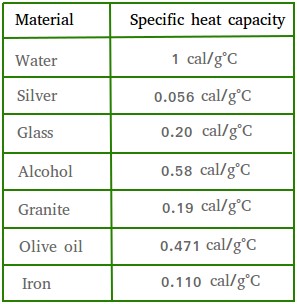
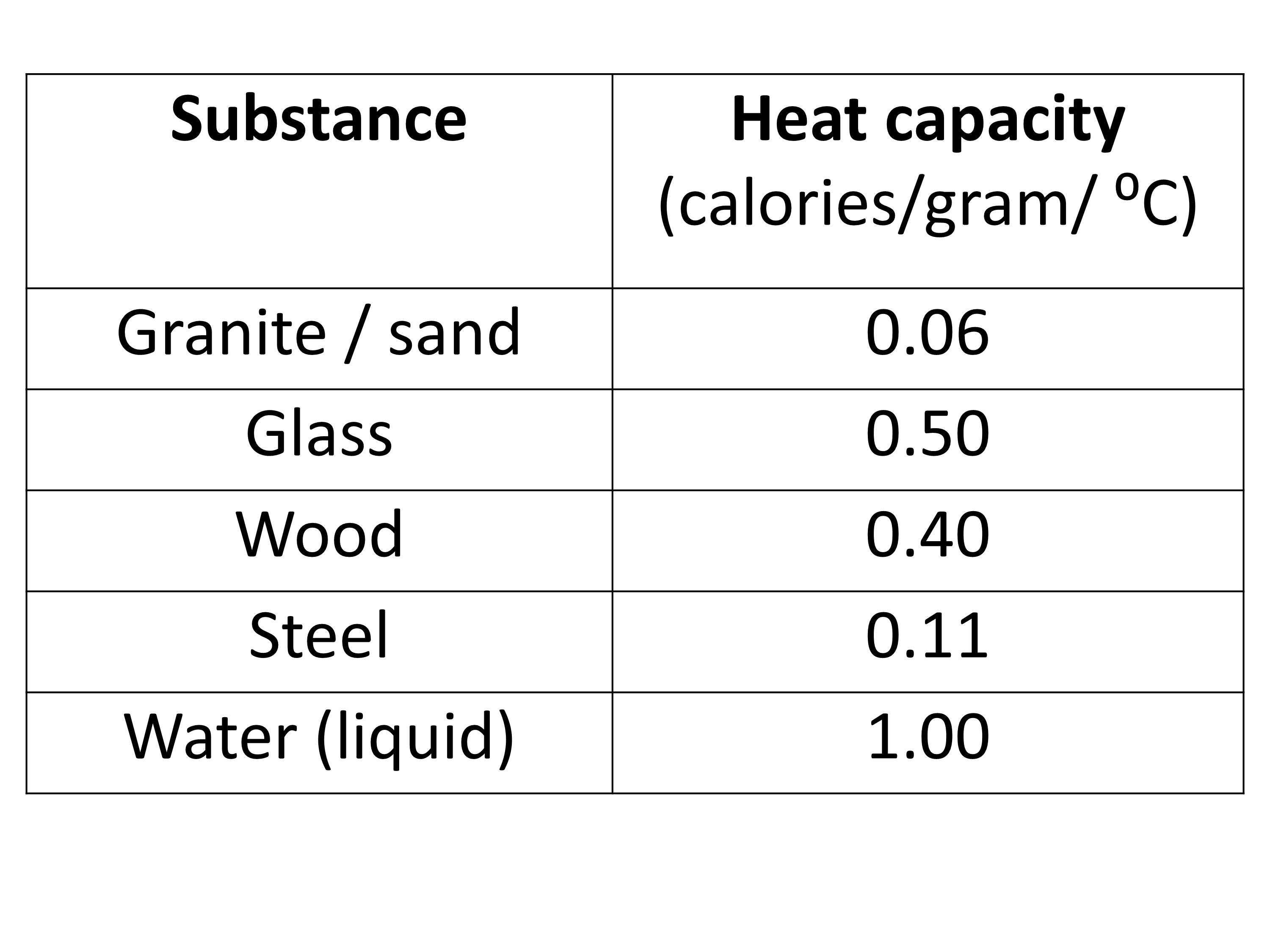

.jpg)
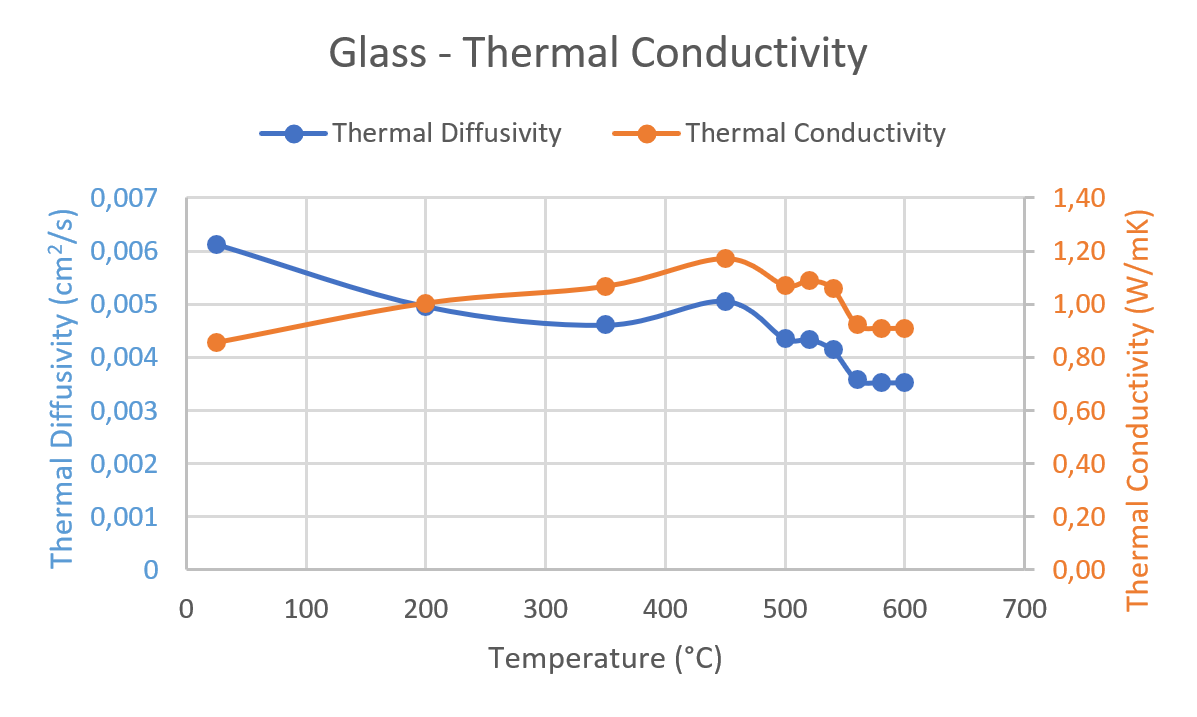



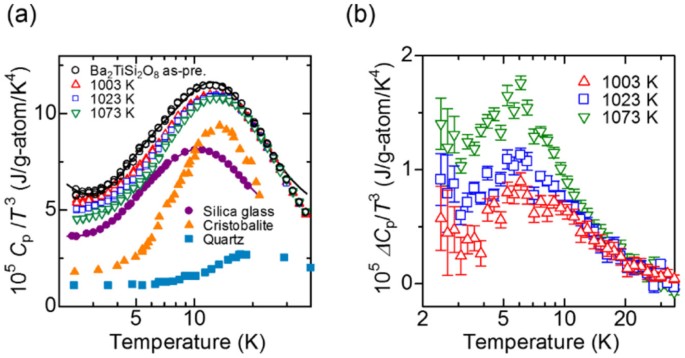
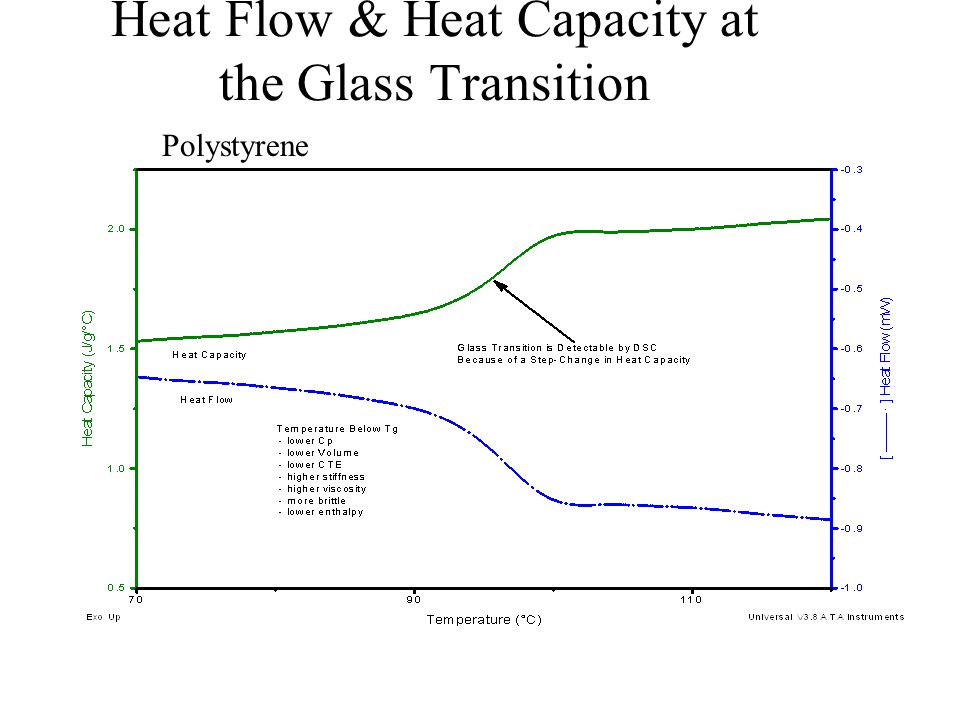

![PDF] Heat capacity at the glass transition | Semantic Scholar PDF] Heat capacity at the glass transition | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ee44b16b7fd93929389afe67bfaf1a6bd994e5b3/5-TableI-1.png)