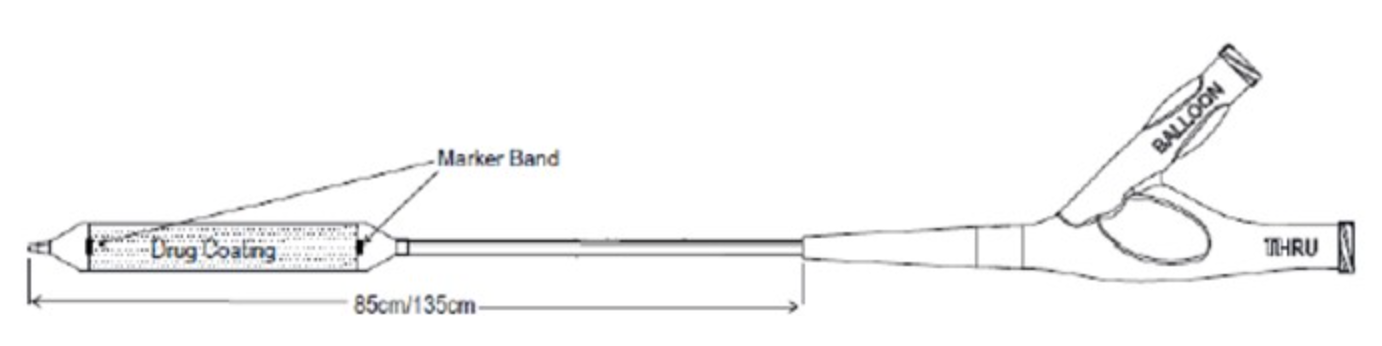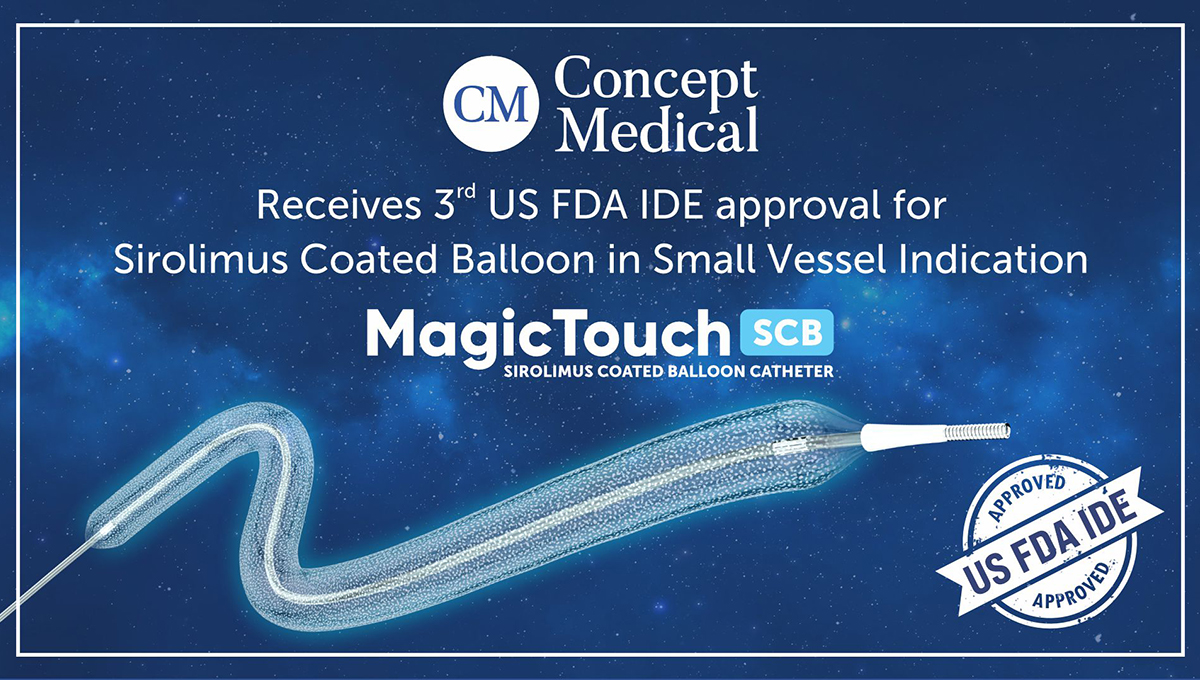
Concept Medical received IDE approval to investigate safety and efficacy of its MagicTouch Sirolimus Coated Balloon Catheter for the treatment of small coronary artery disease | tctmd.com

Chocolate Touch drug-coated angioplasty balloon for treatment of PAD receives FDA approval - Vascular News

FDA deems Orchestra BioMed's sirolimus-eluting balloon a breakthrough device for coronary restenosis | Fierce Biotech

Drug-Coated Balloons for Coronary Artery Disease: Third Report of the International DCB Consensus Group - ScienceDirect

FDA Approves Bard's Lutonix 035 DCB For Dysfunctional AV Fistulae Treatment | Medical Product Outsourcing

MedAlliance's SELUTION SLR drug-eluting balloon (DEB) receives FDA investigational device exemption (IDE) approval, making it the first limus DEB to be available to US patients
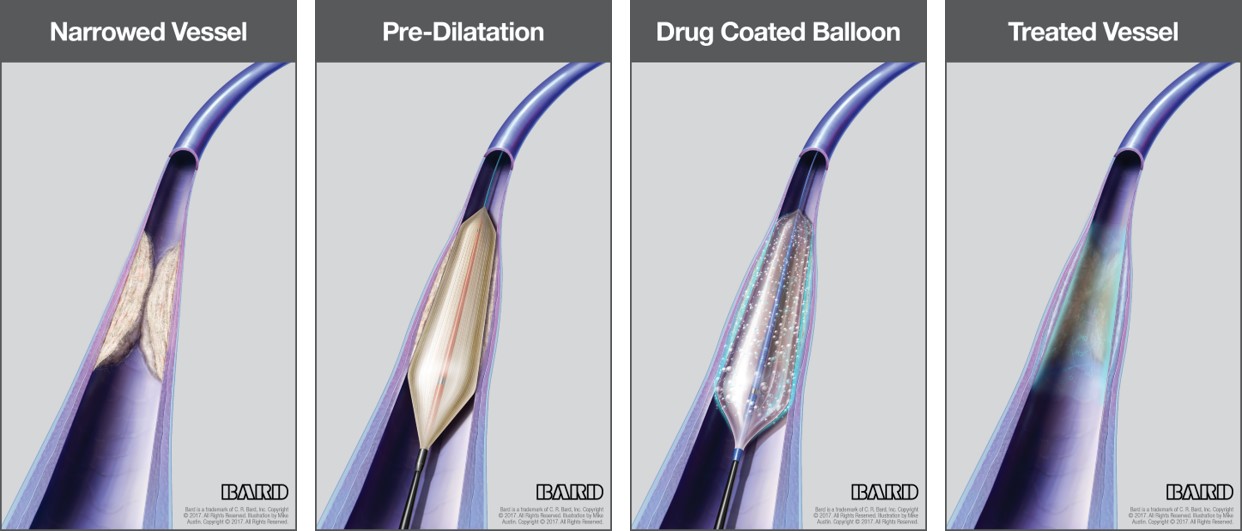
C. R. Bard Receives FDA Premarket Approval For The LUTONIX 035 Drug Coated Balloon - Medical Design and Outsourcing

Concept Medical Granted FDA Breakthrough Device Designation for MagicTouch PTA Sirolimus Coated Balloon | DAIC

FDA panel votes against BD Lutonix drug-coated balloon for below-the-knee - Medical Tubing and Extrusion

Benefit and risk from paclitaxel-coated balloon angioplasty for the treatment of femoropopliteal artery disease: A systematic review and meta-analysis of randomised controlled trials - eClinicalMedicine

Chinese FDA approve Restore drug-coated balloon for in-stent restenosis and small vessel disease – BIBA Medtech Insights


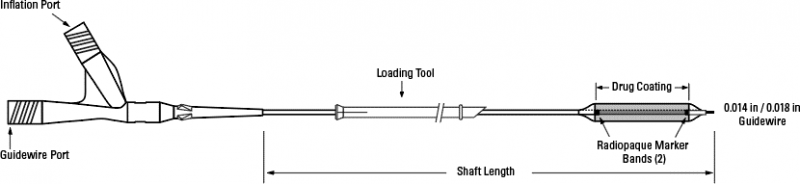

_1653668541.jpg)