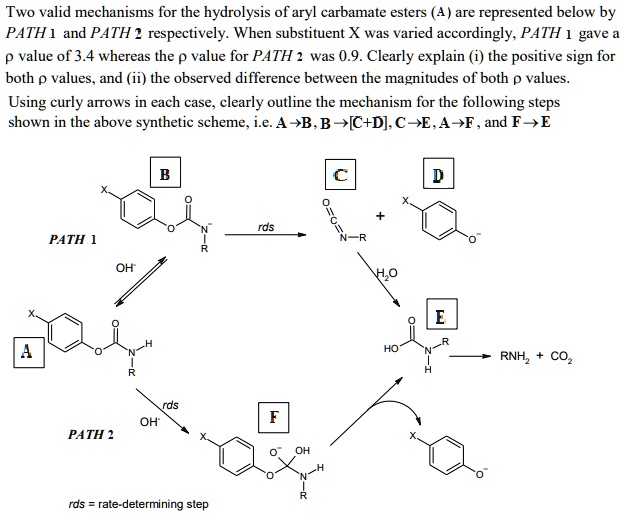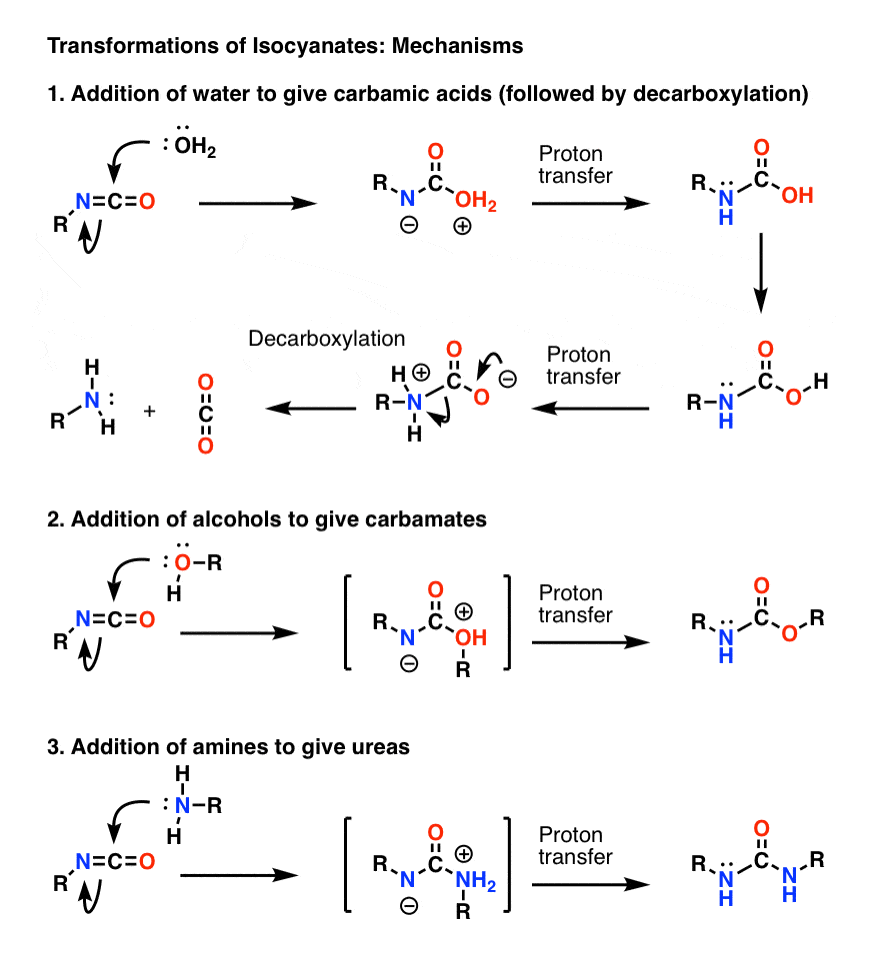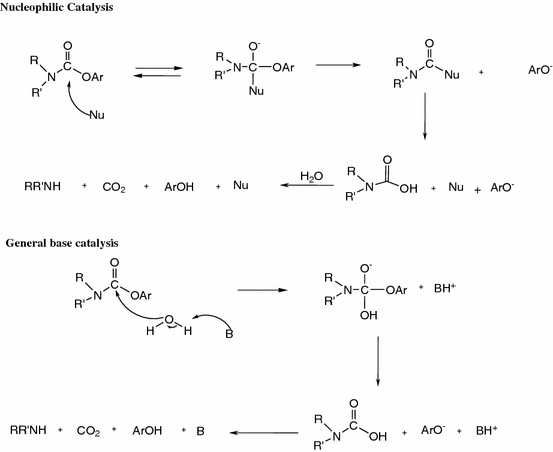
Alkaline hydrolysis of tertiary N-(2-pyridyl)carbamates. Contradictory evidence between nucleophilic and general base catalysis | SpringerLink
Relative Stability of Formamidine and Carbamate Groups in the Bifunctional Pesticide Formetanate Hydrochloride

Triazene drug metabolites. Part 17: synthesis and plasma hydrolysis of acyloxymethyl carbamate derivatives of antitumour triazenes - ScienceDirect

SciELO - Brasil - Kinetics and mechanism of hydrolysis of benzimidazolylcarbamates Kinetics and mechanism of hydrolysis of benzimidazolylcarbamates

Mechanism for the carbaryl hydrolysis and 1-naphthol radical scavenging... | Download Scientific Diagram

Hydrolysis is the most commonly encountered drug degradation mechanism, both in solution and in the solid state. Use the structure of ethyl ethanoate below to illustrate the mechanism of acid-catalyzed hydrolysis of

Hydrolysis susceptibility and carbamate formation for a low moisture-absorbing, siloxane-modified cyanate ester resin matrix (TC410) material used for composite space applications - Rafael J Zaldivar, Geena L Ferrelli, Hyun I Kim, 2022
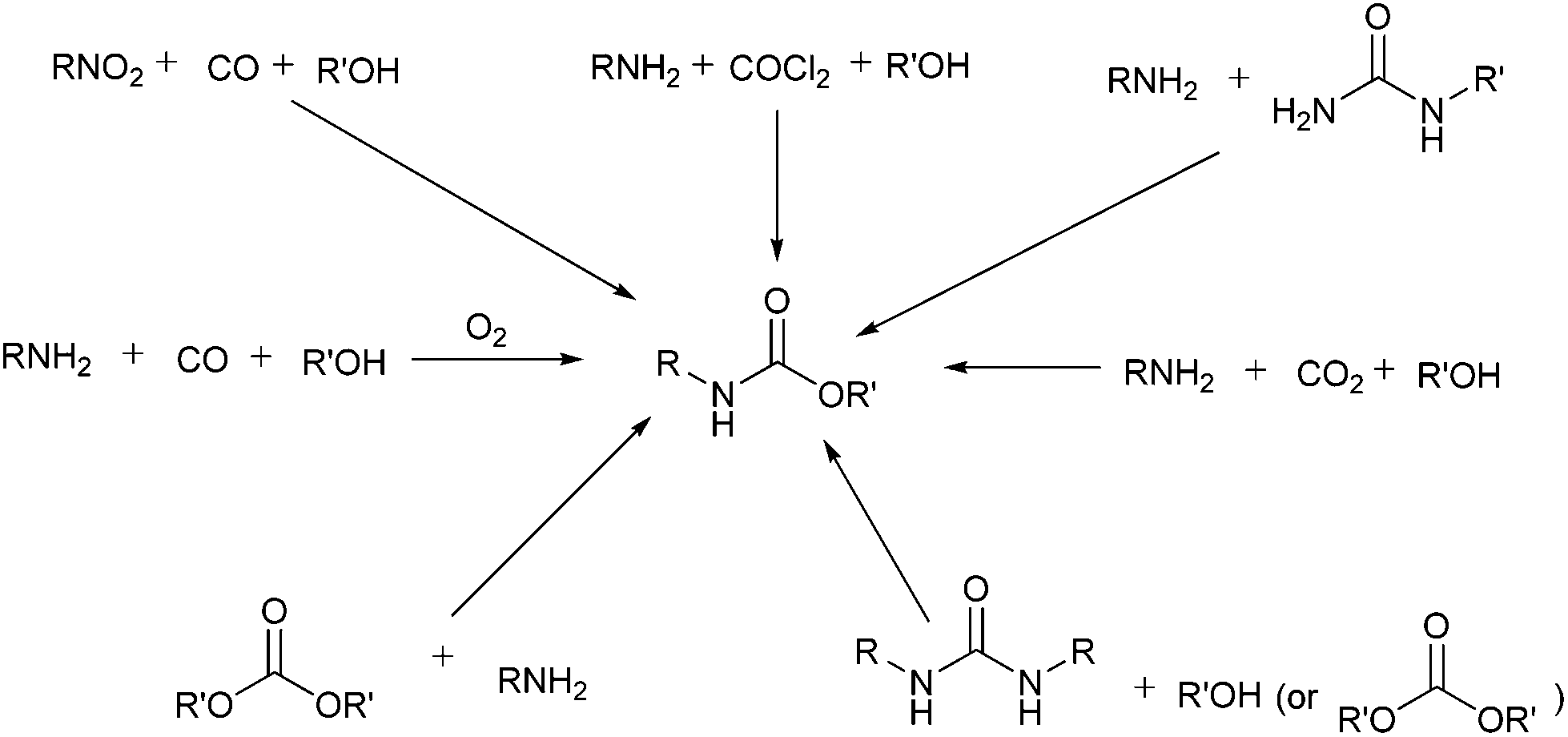
N -Substituted carbamate synthesis using urea as carbonyl source over TiO 2 –Cr 2 O 3 /SiO 2 catalyst - Green Chemistry (RSC Publishing) DOI:10.1039/C5GC01007A

Hydrolysis mechanism of esterases and amidases toward carbamate pesticides | Download Scientific Diagram
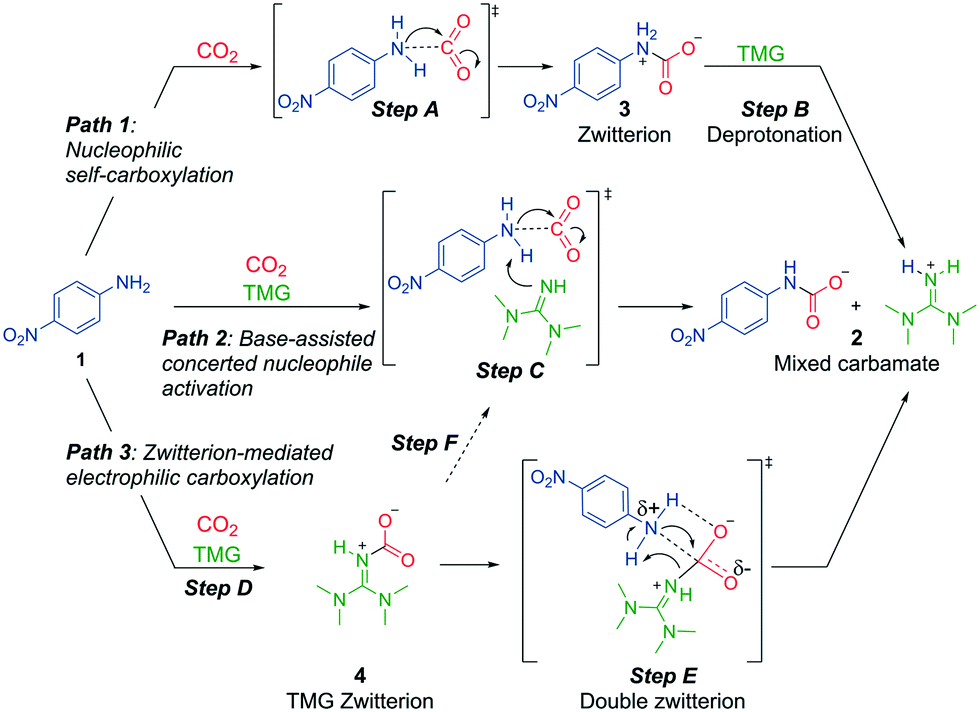
Mechanistic insights into carbamate formation from CO 2 and amines: the role of guanidine–CO 2 adducts - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/D1CY01433A

Alkaline hydrolysis of tertiary N-(2-pyridyl)carbamates. Contradictory evidence between nucleophilic and general base catalysis | SpringerLink